Track & Trace
Government regulation applied to the pharmaceutical industries that requires the implementation of systems tools and artificial vision equipment, to guarantee the authenticity of the medicines produced and commercialized, from the laboratory until their acquisition by the customer.

What is serialization?
Serialization means assigning a unique random serial number to a sales unit so that it is traceable throughout the supply chain according to the GS1® standard.
Control in real time the drug path and location
Follow up the establishments that commercialize the product.
Reduce falsification or adulteration and illegal drug trading
Reduce financial fraud.
Facilitates product recall.
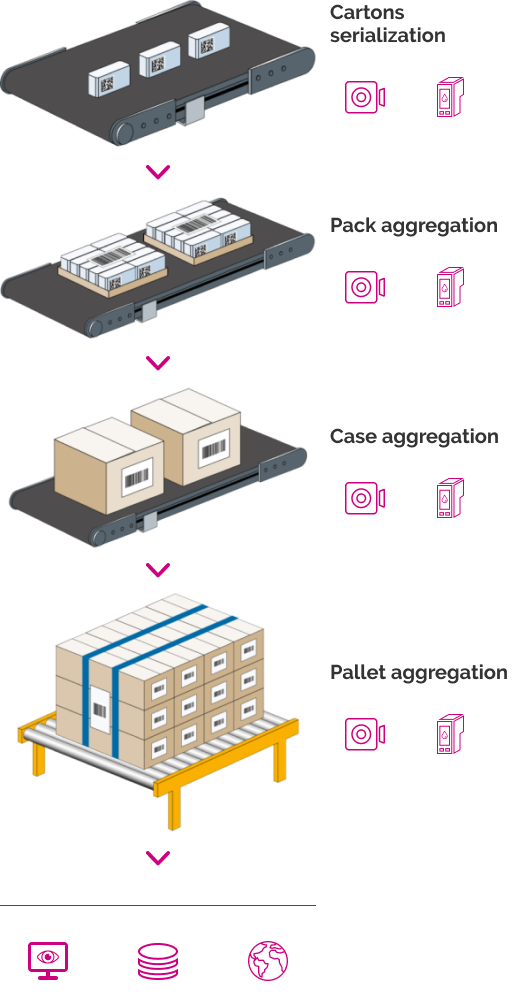
Solutions for Track & Trace automatization in manual or automatic packing lines
Cartons Serialization
- Stand Alone equipment for on-line or off-line operations.
- Cartons printing: Datamatrix, GTIN, Serial No., Expiration, Lot.
- Datamatrix and alphanumeric serialization of primary or secondary packaging in high speed line (400ppm).
- Datamatrix according to ISO 15415 and OCR/OCV of alphanumeric characters.
- Discard control for non-conforming units.
Aggregation control in boxes, packs and pallets:
- Sequential or simultaneous reading of unique codes within a box or pack.
- Printing and application of unique labels for packs or boxes.
- Establishment of parent-child relationship through real-time linkage with production line equipment (Level 2) at unit, pack or shipping carton, pallet level.
Software solutions:
- Easy to use software, user friendly interface and virtual voice assistance for quick setups.
- Level 3 software solutions for serial number generation and batch management brought in pharmaceutical laboratories.
- Functionalities specifically designed for the industry: sample withdrawal, quarantine lots, interaction with sanitary authority, etc.
- Specific development of interfaces for data exchange with ERP, MES, WMS.
- Application for tracking units throughout the distribution chain: drugstores, distributors, logistics operators, hospitals and pharmacies.
- Compliance with FDA 21CFR Part 11: electronic signature by biometric identification, and change audit protected by encryption.
- Scalability of systems for Industry 4.0.
- Suitable for integration in new and used machines.
Compact carton serialization module for traceability, integrating printer, inspection camera and reject in a self-contained transport unit.
Small size, suitable for line integration or as a stand-alone manual feeding module.
The equipment interacts with the track & trace system, receiving the batch serial numbers or generating them by itself.
Controls different types of common encoders on the market according to ISO 15415 to print the data on the carton: Datamatrix, GTIN, Serial No., Lot, Expiration, Fabrication, etc.
Updates the information validated in the track & trace management system database: codes, printed, verified, discarded, seized, etc.
Quick and easy format adjustment.
Serialization process
Serialization equipment specially designed for the pharmaceutical industry, integrated with Stand Alone tapes, labelers, Tamper Evident applicators and dynamic scales according to project.

Aggregation



Module for end-of-line Track & Trace control, designed to associate the Datamatrix codes of the cartons with the grouping label (pack, case and pallet).
It performs simultaneous reading of the cases inside a pack or box through a single image or through sequential reading on the machine in special cases.
The equipment interacts with the traceability system by receiving the serial numbers printed on the lot cases or it can build a base by itself based on the reading of the inspected datamatrix.
Generates serialized pack, case and pallet grouping labels and updates the database with the corresponding serial numbers.
Alerts on the presence of products that do not meet the printing quality requirements and/or have erroneous codes.
Compact and modular equipment capable of being integrated into existing lines or Stand Alone modules.
Quick and easy format adjustment.
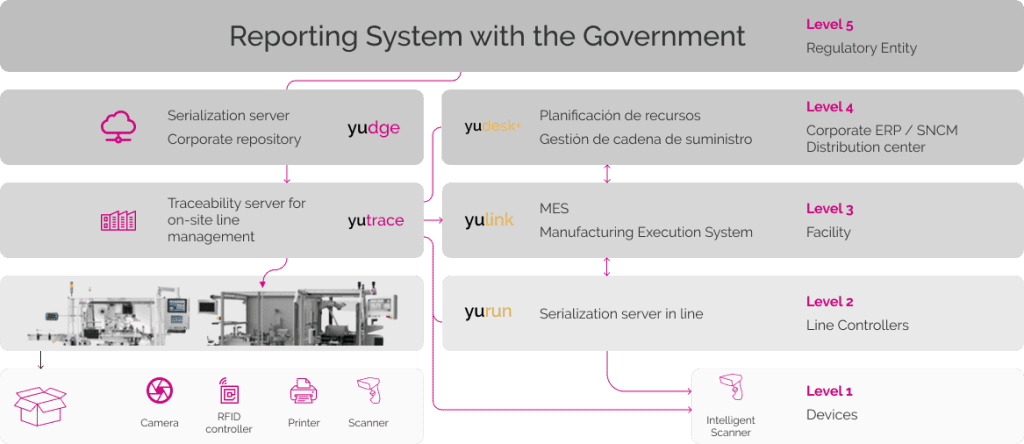
Modular and Scalable System Solution Level 3
(according to ISA 95)
Project management : A comprehensive solution, turnkey solution
Our advantages : Flexible software
Modularity
Customizable
Low impact on validation
Integration with ERPS
Integration with approved L4
Modular and scalable solution to minimize the initial investment and guarantee the continuity of the business in the future.
Lists and demonstrable integrations in association with all nationally and internationally recognized Level 4 providers.
Automation of production orders and master data from integration with ERP / MES.
Integración en asociación con OEM reconocidos internacional y nacionalmente.
Efficient integration into existing machines, minimizing implementation time.
SLA tailor-made customer. Warranty on local supplies for 10 years.
Case Conveyor Module
Stand Alone equipment capable of integrating printer and control system for serialization or OCR/OCV control.
The module has a lower and upper band to guarantee the stability of the case, achieving precision in the printing of alphanumeric characters and 2D codes, and the corresponding inspection and discarding of these (OCR/ Grading ISO 15415).
This system is designed to meet the requirements and regulations of the pharmaceutical industry and can be integrated into the existing production line by adapting to upstream and downstream machines, as well as off-line with manual feeding.
Technical Characteristics:
- Aluminum conveyor with APM (anti-wear) sliding track and double polyurethane belt (upper and lower), driven by a stepper motor.
- Speed up to 250 cartons / minute.
- Industrial PLC used for system management and connectivity with adjacent machines.
- Precision regulators with analog display for standardizing the setup of the different cartons formats.
- Adjustable precision regulator supports for side guides, vision camera and printers (CIJ, TIJ, Laser, others).
- AISI 304 stainless steel structure and height adjustable legs (mobile option with wheels upon request).
- Adjustable height (800~900mm)
- Adjustable speed from HMI screen.
- Pneumatically operated ejection system with FESTO solenoid valve and sensors for reject confirmation.
- Stainless steel waste drawer with lockable door.
- Emergency push button, reset button and dedicated safety relay.

A reliable solution prepared for the future
Native integration with yudoo
Track & Trace Solution is natively integrated with Yudoo, the 4.0 pharmaceutical software suite for the full management of automation processes, digital quality, data analysis and for Track&Trace. A digital hub that is everywhere accessible through a secure-access web interface or dedicated devices.
A scalable and modular suite, including tools for automation (centralized management of production, workflows and timesheet), for the digital quality (creation of paperless systems, support systems for line clearance operations), for data analysis (analysis of production data, business intelligence dashboard, condition monitoring and predictive maintenance) and for Track and Trace (complete solutions for Level 3 and 4 of serialization, monitoring of serialization operations).
Yudoo offers multiple advantages such as the possibility to centralize production formats and data, avoiding duplicates and reducing work time. The suite can connect to existing company systems to retrieve updated data exactly from where they are.
