Vision systems, track and trace and 4.0 software suite.
Discover here SEA Vision solutions dedicated to laboratories and manufacturers of pharmaceutical packaging machinery.

Discover here SEA Vision solutions dedicated to laboratories and manufacturers of pharmaceutical packaging machinery.


Our equipments provide a wide variety of solutions to control the quality of pharmaceutical packaging. Developed according to the highest quality standards, our equipment offers several advantages and tools to simplify the operation.
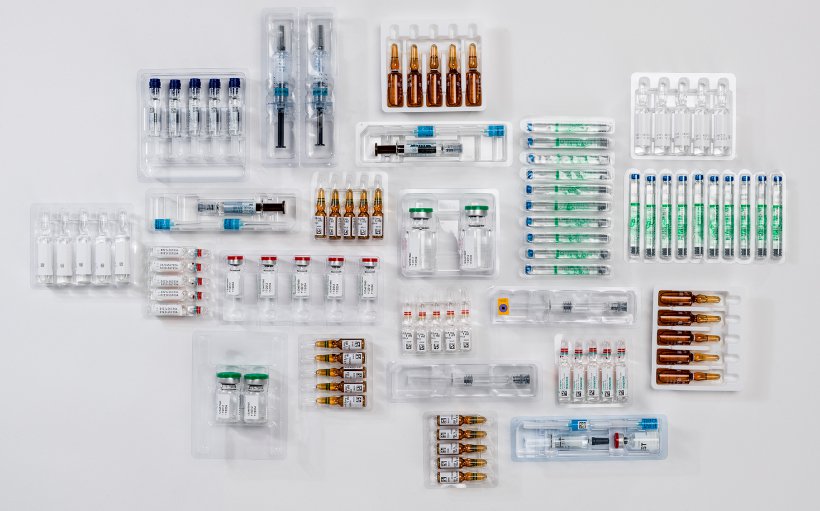
Our vision systems ensure compliance with the quality standards required by pharmaceutical industry regulations and allow control activities during all phases of drug production and packaging. They also have a series of independent manual stations with an integrated vision system to perform special drug checks before placing the products on the production lines.
In recent years, regulations on the serialization and traceability of medicines have assumed an increasingly important role in a growing number of countries.
Nowadays, SEA Vision has over 1,800 traceability system installations on a wide variety of machines, giving pharmaceutical laboratories what they need to comply with anti-counterfeiting guidelines.


According to the regulations of each market, our technology allows us to track each piece throughout the supply chain and thus guarantee the origin and authenticity of the product.
Our software allows:

If during the 3.0 industrial revolution – that of automation – companies invested to make production lines efficient and automatic, today the 4.0 challenge requires equipping themselves with digital tools to centralize production management in order to improve processes, transform existing technologies and make data-driven decisions.
But how to integrate new 4.0 tools into an existing technological context, strictly connected to consolidated and compliant production processes and procedures?

Our vision systems ensure compliance with the quality standards required by pharmaceutical industry regulations and allow control activities during all phases of drug production and packaging. They also have a series of independent manual stations with an integrated vision system to perform special drug checks before placing the products on the production lines.
According to the regulations of each market, our technology allows us to track each piece throughout the supply chain and thus guarantee the origin and authenticity of the product.
Our software allows:

Our vision systems ensure compliance with the quality standards required by pharmaceutical industry regulations and allow control activities during all phases of drug production and packaging. They also have a series of independent manual stations with an integrated vision system to perform special drug checks before placing the products on the production lines.

Yudoo comes from a consolidated and long experience in automation and software integration management for the pharmaceutical industry. This platform allows integration with existing systems (such as ERP or MES and others) to manage production and T&T processes from a centralized platform, collecting and transforming data into information.
By analyzing state-of-the-art algorithms and powerful Business Intelligence functionalities, yudoo is able to reveal preventive and predictive insights to answer any questions that may arise.